FDA Approves Long-Acting Medication for People With Multidrug-Resistant HIV
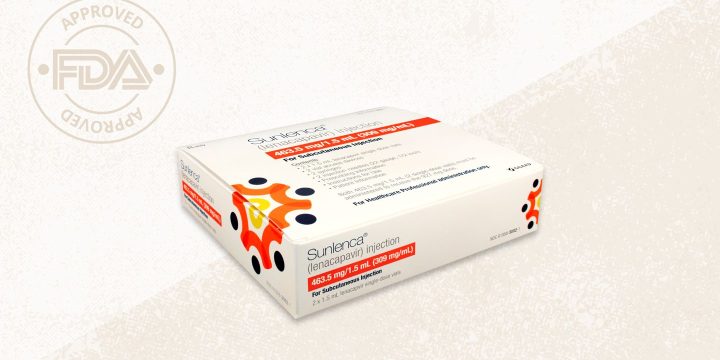
The U.S. Food and Drug Administration (FDA) has approved Sunlenca (lenacapavir), a new type of medication for the most common variant of HIV, HIV-1 (human immunodeficiency virus type 1).Lenacapavir is designed for heavily treatment-experienced adults with multidrug-resistant HIV who are not able to adequately manage the virus with their current treatment regimen. It is an injectable medication administered under the skin (subcutaneously) once every six months.Developed by Gilead Sciences, lenacapavir is the first of a new class of HIV medications called capsid inhibitors, which work by blocking the virus’s protein shell (the capsid), thereby interfering with essential steps of the virus’s life cycle.In a clinical trial, lenacapavir was able to lower the viral load in patients who were not responding adequately to other therapies.“Today’s approval ushers in a new class…