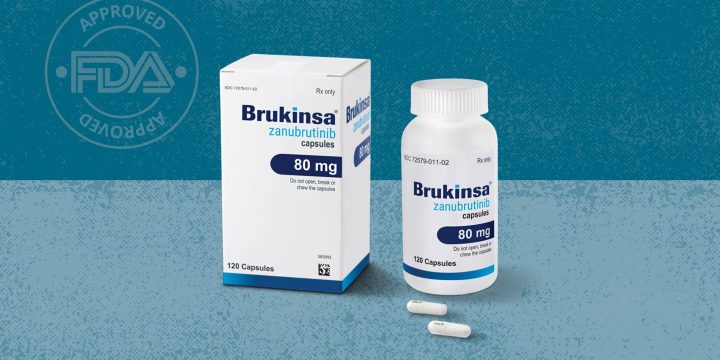
FDA Approves Brukinsa (Zanubrutinib) for Leukemia and Lymphoma
The U.S. Food and Drug Administration (FDA) has approved Brukinsa (zanubrutinib) for the treatment of the blood and bone cancers known as chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL). The National Comprehensive Cancer Network has endorsed zanubrutinib as a CLL-SLL treatment for both newly diagnosed and previously treated patients.Zanubrutinib belongs to a class of drugs known as BTK inhibitors (BTKi), which work by preventing the growth of malignant B-cells (white blood cells that make illness-fighting antibodies), according to the National Cancer Institute.The FDA previously approved the drug, which is made by the Beijing-based pharmaceutical company BeiGene, for three other forms of lymphatic cancer: Waldenström’s macroglobulinemia, marginal zone lymphoma, and mantle cell lymphoma.CLL is a common form of leukemia, accounting for about one-quarter of all new cases each year; the American…