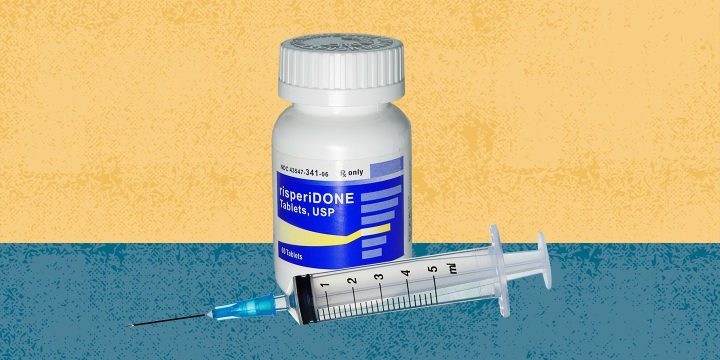
FDA Approves Rykindo, a Long-Lasting Injectable, for Schizophrenia and Bipolar I
Men and women who take risperidone to treat schizophrenia or bipolar I have a new option if they aren’t able to take oral medications every day or prefer a long-lasting alternative. The U.S. Food and Drug Administration (FDA) approved Rykindo as an extended-release injectable version of risperidone on January 15, 2023, according to a press release from Luye Pharma, the company that developed the drug.Rykindo is administered via intramuscular injection once every two weeks, delivering risperidone via long-acting and extended-release “microsphere” technology. These microspheres release medication at a gradual, steady rate. The injection needs to be administered by a healthcare professional.Risperidone Has Long Been Used to Treat Schizophrenia and Bipolar IRisperidone was first approved by the FDA in 1993 to treat symptoms of schizophrenia, according to StatPearls. A long-acting injectable form of…





