FDA Approves Leqembi, New Treatment for Early-Stage Alzheimer’s Disease
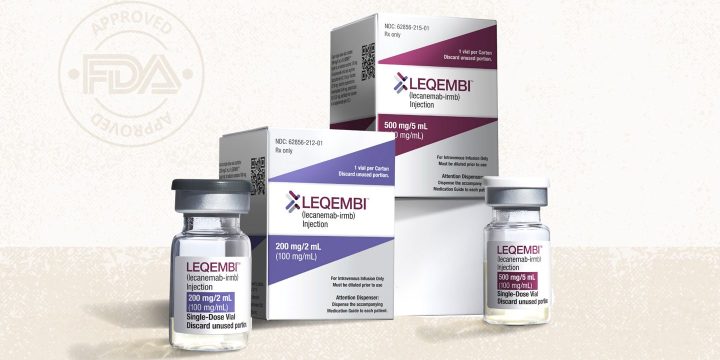
On Friday, January 6, the U.S. Food and Drug Administration (FDA) granted accelerated approval of a new drug, Leqembi (lecanemab-irmb), for the treatment of early-stage Alzheimer’s disease.Leqembi works by removing amyloid beta plaques, a protein that can accumulate in the brains of people with Alzheimer’s and that is believed to be a key factor in the disease.According to the FDA, Leqembi was evaluated for the accelerated approval in a double-blind, placebo-controlled trial that included 856 people with Alzheimer’s disease. The medication, given every two weeks as an intravenous infusion, was given to subjects with mild cognitive impairment who also had imaging scans to confirm amyloid plaques in their brains.Data submitted by Eisai — the company that makes Leqembi — to the FDA shows that patients who received Leqembi had significant…