High Blood Pressure Medicine Recalled Again Due to Possible Cancer Risk
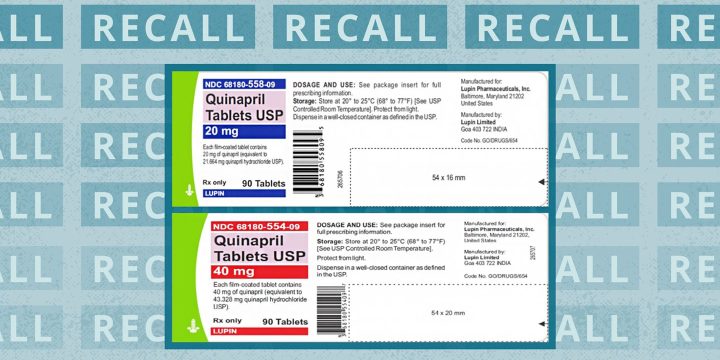
Four lots of the generic high blood pressure medication quinapril tablets have been recalled by its manufacturer, Lupin Pharmaceuticals, because of abnormally high amounts of a probable cancer-causing impurity.Recent testing revealed levels of a nitrosamine impurity (N-nitroso-quinapril) were above the U.S. Food and Drug Administration's (FDA) acceptable daily intake level.While long-term ingestion of this suspected carcinogen may increase cancer risk, there is no immediate danger to patients taking this medication.According to an announcement from the FDA issued on December 21, the quinapril products in question are:20 mg tablets, lot number G102929, expiration date 04/202340 mg tablets, lot number G100533, expiration date 12/202240 mg tablets, lot number G100534, expiration date 12/202240 mg tablets, lot number G203071, expiration date 03/2024The pills were packaged in 90-count bottles and distributed nationwide to wholesalers, drug chains, mail…